Article topic: Radiotherapy Techniques
Author: Saif Yahya Smadi
Editor: Dr.Omar Jbarah, Philip Sweidan
Keywords: Intensity-modulated therapy (IMRT), Brachytherapy, Stereotactic body radiotherapy (SBRT), Stereotactic radiosurgery (SRS), Charged particle therapy.
Overview
Cancer is a worldwide public health problem; it accounts for a quarter of deaths exceeding heart diseases as the leading cause of death for people under 85 years old. Radiotherapy is an important tool in the management of cancer. More than 50% of cancer patients with localized tumors receive radiotherapy as part of their therapy, either alone or combined with other types of management such as chemotherapy and surgery(1). In this article, we are going to briefly discuss some radiotherapy techniques.
Intensity-modulated radiotherapy
Intensity-modulated radiotherapy (IMRT) is a radiotherapy treatment that provides a high dose of radiation to the tissue affecting the tumor but with minimal dose to the nearby normal tissue. There is an obvious relationship between tumor control and radiation dose, but the radiation dose is usually limited by the tolerance of the surrounding structures. In some cases the use of this technique allows more normal tissue to survive than other techniques(2).
IMRT treatment plans are produced using an inverse planning system that uses computerized techniques to distribute the intensities across the target volume(3). Therefore, this technique is found to be appropriate for head and neck cancers which are located near many critical organs as the brainstem, eyes, spinal cord, optic nerves, lacrimal glands, cochlea, and mandible(4)(5). A newly published systematic review identified 10 comparative studies of IMRT and 2D-RT in nasopharyngeal carcinoma, they concluded that IMRT yields a lower incidence of late toxicities and thus it may be an effective treatment for patients with nasopharyngeal carcinoma(6).
This technique improved the target conformity, increased the normal tissue sparing, and enabled dose escalation. On the other hand, the biggest downside of IMRT is the increase in total body irradiation dose, in addition to that, it is a complex and time-consuming procedure that requires a long time, this long time may interfere with the patient’s comfort and positioning(2)(7).
Brachytherapy
Brachytherapy is a particular type of radiation therapy that requires the placement of an emitting radiation source close to the tumor or adjacent tissue at risk of having the microscopic disease. Brachytherapy applicators are specifically designed for different anatomic sites like body cavities and tissues(8).
Implanted radon (Rn) in the brain of a patient who was suffering from a pituitary mass was the first use of brachytherapy for brain tumors by Dr. W.O Lodge in 1936(9).Iodine-125 brachytherapy is an Important therapeutic method that enhances the survival of patients with gliomas(10)(11). Recently, it has been suggested that the use of I125brachytherapy be effective in patients with recurrent gliomas(12)(13). A new study published in 2020 states that I125 brachytherapy could be an effective method in recurrent high-grade gliomas (HGG) even without the use of radiotherapy and/or chemotherapy(14).
The main limitation of this procedure is that it is invasive, meaning that it requires a short procedure in the operating room to place the source. Another important aspect is the quality of the implantation. Certainly, inappropriate placement of the source may expose the tumor to under dosage or too much toxicity especially if the source is close to a critical organ(15).
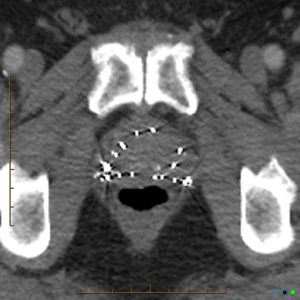
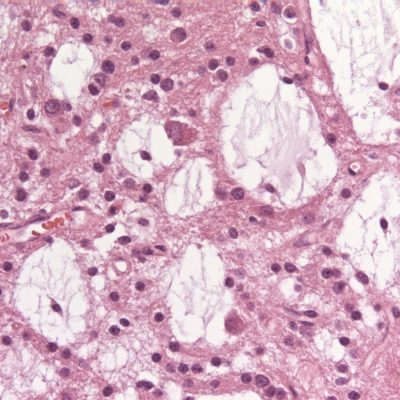
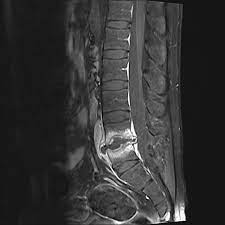
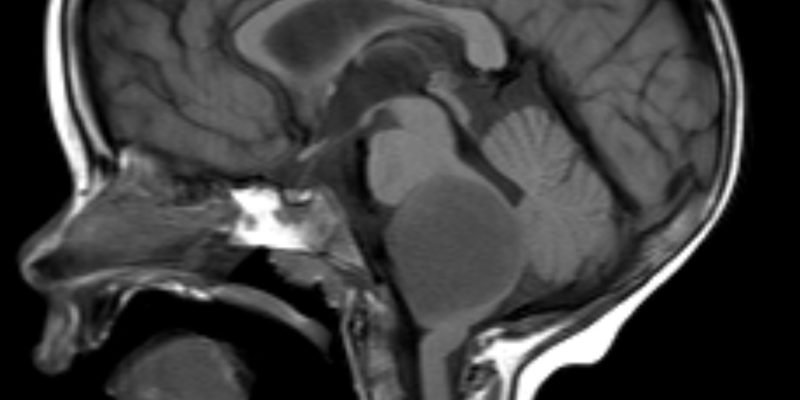
Figure1: prostatic brachytherapy, Case courtesy of Dr Chris O’Donnell(32).
Stereotactic body radiotherapy
Stereotactic body radiotherapy (SBRT) is a radiation method of delivering a high dose of external radiotherapy beam to small tumor targets very precisely with a high level of accuracy(16). Stereotactic body radiotherapy development began in the 1990s in Stockholm at Karolinska Institute(17).
Compared with other external beam radiation therapies, this technique has many advantages, the use of high dose radiation, the delivery of one to five fractions in a few days, decreasing the treatment overall time, and improving the treatment response(18). On the other hand, missing the target tumor can cause damage to the surrounding normal tissues, and because SBRT radiation fraction is higher in dose compared to other types of external beam radiation therapies the potential damage is also higher(19).
Stereotactic radiosurgery
Stereotactic radiosurgery (SRS) was first introduced in 1951 by Lars Leksell as an alternative for whole-brain radiotherapy(20)(21). SRS uses multiple beams of protons, gamma rays, and high-energy x-rays(22). This technique works by radiation-induced DNA damage by free radicles and ions(23).
It is a non-invasive, outpatient procedure that does not require general anesthesia. Under local anesthesia, a special helmet produces gamma rays fixed to the patient’s skull(23). Stereotactic radiosurgery (SRS) is considered suitable for brain metastasis and primary tumors, such as vestibular schwannomas, meningiomas, pituitary tumors, and other tumors that are resistant to conventional whole-brain radiation therapy (WBRT)(24)(25).
The use of this technique is contraindicated in large lesions or too many lesions, the single-fraction SRS is often limited to small lesions(26). A randomized trial compared SRS followed by conventional radiotherapy with carmustine (chemotherapy) versus only conventional radiotherapy with carmustine for patients with glioblastoma showed no improvement in the outcome or the general quality of life(27). 
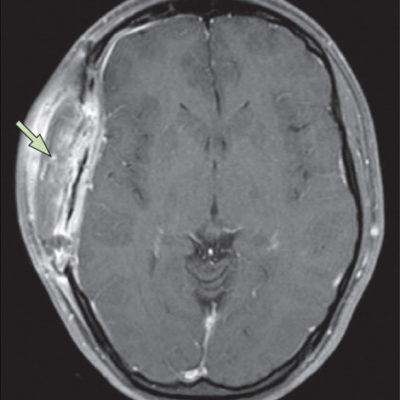
Figure2: Brainstem glioma, Case courtesy of Assoc Prof Frank Gaillard(33).
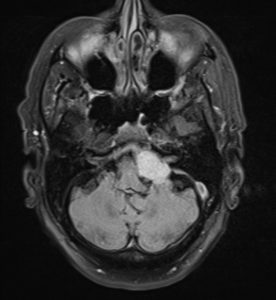
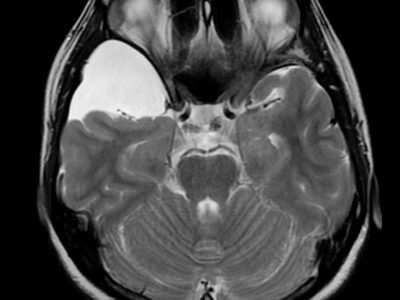
Figure3: Cerebellopontine angle meningioma, Case courtesy of Dr Mostafa El-Feky(28).
Charged particle therapy
Robert R.Wilson was the first one who proposed that accelerated protons could be used in the treatment of localized tumors in 1946. In 1950 was the first clinical use of this technique for pituitary hormone suppression in metastatic breast carcinoma(29).
Unlike X-Ray irradiation, which is absorbed by the body and shows an exponential decrease in the radiation dose with increasing tissue depth, this technique deposits little energy at the body’s surface(30). Therefore, charged particle therapy produces the same dose of radiation to a tumor, this will minimize the radiation received by nearby normal nonmalignant tissues(31).
Charged particle therapy is useful in treating tumors surrounded by radiosensitive tissues and in situations where surgical access is limited. A small number of studies have made comparisons between X-rays and charged particle therapy. Although there are supporting advantages for charged particle therapy the cost-benefit ratio remains debatable(30).
External Links:
Brachytherapy
https://www.youtube.com/watch?v=GfuFgyULt7w
Stereotactic radiosurgery
https://www.youtube.com/watch?v=HZQ8puGlsIw
Charged particle therapy