Author: Zaid Sayegh
Editors: Miramar Haddad, Haneen A. Banihani
Reviewer: Dr Adam M. Abdallah
Introduction
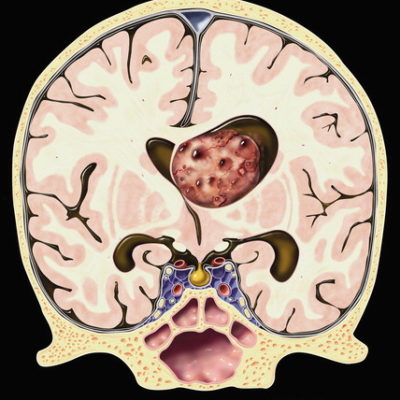
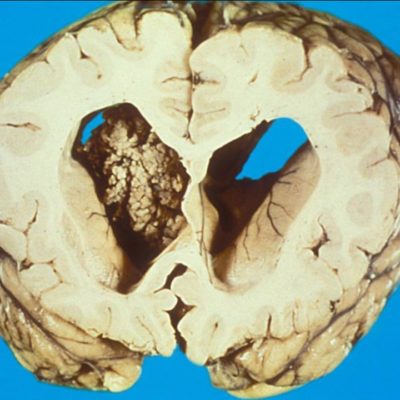
Neuro-oncology is a rapidly evolving subspecialty in medicine that tackles primary or metastatic CNS and PNS neoplasms and complications concerning the nervous system occurring either directly or indirectly due to nervous system or systemic malignancies.[1] There are many distinct types of brain tumors, which are divided into benign and malignant. Benign tumors are usually slow-growing and do not invade nearby tissues, and the most common type is meningiomas, followed by pituitary and nerve sheath tumors. Conversely, malignant neoplasms grow quickly, uncontrollably, and infiltrate other tissues, which explains why malignant brain neoplasms are classified into primary tumors (originating from the brain like gliomas) and metastases (originating from different regions in the body).[2,3] Around 80% of malignant brain neoplasms are gliomas originating from glial cells, specifically astrocytes, oligodendrocytes, and ependymal cells. Gliomas can either be “diffuse gliomas,” which show extensive invasive growth, or “non-diffuse gliomas,” which are circumscribed. Diffuse gliomas fall into three subdivisions: diffuse astrocytomas, oligodendrogliomas, and oligoastrocytomas. Within these subdivisions, a grade is typically assigned: grade II (low grade) is characterized by nuclear atypia, grade III shows increased mitotic activity, and grade IV is characterized by additional microvascular proliferation, necrosis, or other molecular findings such as the presence of a CDKN2A/B homozygous deletion that causes a worse prognosis[3]
The 2021 WHO Classification of CNS Tumors
In the past, the classification of CNS tumors had been based on histological findings with supplementary tissue-based tests like immunohistochemical or ultrastructural. Over the past few years, molecular biomarkers have been given more importance and therefore employed in the diagnostic process of tumors.
In the context of taxonomy, “type” is now used instead of “entity”, while “subtype” is used instead of “variant”. Furthermore, some diagnoses have been redefined and some terms like “anaplastic” have been removed consequently leading to some diagnoses being removed such as “anaplastic astrocytoma” and “anaplastic oligodendroglioma” (“anaplastic meningioma” was retained). For some tumors like choroid glioma, the anatomical site (of the third ventricle) was omitted.
Furthermore, when reporting gene symbols/names or chromosome alterations, WHO CNS5 has standardized the structure; gene symbols are italicized whereas proteins and gene groups are not. In addition to that, the size of the tumor is now reported in millimeters (mm) instead of centimeters (cm) to eliminate the usage of decimal points.
In terms of tumor grading, WHO CNS5 has altered two aspects: Roman numerals have been replaced by Arabic numerals and tumors are graded within types instead of across different tumor types. For instance, the old WHO classification labeled an anaplastic astrocytoma as grade III (the old classification used Roman numerals), the same grade as an anaplastic meningioma even if they are biologically unrelated. The idea was that same-grade tumors have approximately similar survival times. Just like how a prostate or breast cancer can be assigned grades 1 to 4, WHO CNS5 extended within-tumor-type grading to many categories. This alteration aimed to enhance the versatility of grade utilization relative to the tumor type, as well as to highlight the biological similarities among tumor types instead of the clinical characteristics, and finally to align with the WHO grading system for non-CNS tumor categories. It is also advised to use the term “CNS WHO grade” when assigning a grade.[4]
Clinicopathological grading
WHO CNS5 has in general maintained the grade ranges used for tumor types. To elaborate, IDH-mutant astrocytomas are still assigned a grade between 2-4 while meningiomas are assigned grades 1-3.
There is some conflict regarding the need to assign a specific grade for a tumor as it could confuse clinical care. For instance, WNT-activated medulloblastoma is an embryonal tumor that has a very aggressive clinical course if left untreated, but since its highly responsive to the current therapeutic protocols, almost all patients have long-term survival. This means that assigning it as CNS WHO grade 4 like the other grade 4 tumors indicates a bad prognosis which is false. On the other hand, assigning it as CNS WHO grade 1 like the other grade 1 tumors gives a false sense that the tumor is benign. [4]
Combined histological and molecular grading
As mentioned before, molecular markers now play a vital role in the prediction of the clinical course and prognosis of a tumor. For example, in IDH-wildtype diffuse astrocytomas, certain mutations like CDKN2A/B homozygous deletion in IDH-mutant astrocytomas, TERT promoter mutation, EGFR amplification, and +7/−10 copy number variation allow the designation of CNS WHO grade 4 even if it histologically should be assigned a lower grade. [4] For further reading, please visit https://tumourclassification.iarc.who.int/
Carcinogenesis
Research done over many years has shed some light on the fundamentals of tumorigenesis and shown that there are two main phases: the preliminary phase, characterized by intracellular alterations, and the phase of clinical presentations.[3] We have also discovered eight hallmarks of cancer that are: self-sufficiency in growth signals, insensitivity to inhibitory growth signals, evasion of apoptosis, limitless replicative potential, sustained angiogenesis, ability to invade and metastasize, reprogramming of metabolism, and evasion of the immune system.[5] Neural stem cells (NSC) display self-renewing properties and resilience, making them highly prone to carcinogenesis. These cells (NSC) typically differentiate into neurons, oligodendrocytes, and astrocytes; therefore, where the mutations happen will dictate the type of tumor that forms. [3]
Imaging
Imaging in neuro-oncology plays a vital role in diagnosing, prognosis, and treating response assessment of central nervous system (CNS) tumors.[6] The current standard radiographic evaluation of neuro-oncologic diseases typically involves the utilization of computed tomography (CT) and magnetic resonance imaging (MRI). Although CT imaging is more rapid and convenient than MRI, it is less sensitive because the images are captured at a higher rate. CT imaging permits remarkable differentiation of hard tissues such as bony or calcified structures, and it is also considered an excellent tool for identifying hemorrhages which can be utilized for a rapid postoperative assessment. Conversely, it is weak at recognizing different tissues, therefore, demanding the use of contrast agents to ameliorate the quality of the images, which might not be feasible in patients with renal disease or allergies to contrast.[7]
MRI with gadolinium contrast is the gold standard for assessing brain tumors before and after therapy. Compared to CT imaging, MRI provides better anatomic resolution without requiring ionizing radiation or iodine contrast medium, making it supreme. Contrast-enhanced T1-weighted imaging is splendid at emphasizing areas of blood-brain barrier breakdown; however, it should be collated with non-contrast–enhanced T1 imaging to discern areas of interest from fat, blood products, and proteinaceous material. On the other hand, T2-weighted imaging is more sensitive to minor changes in the brain’s water content, making it better suited to identify subtle changes in tumor-related edema. Fluid-attenuated inversion recovery (FLAIR) imaging suppresses CSF signal and outlines lesions adjacent to areas rich in CSF [7,8]
MRI software developments have allowed us to utilize this technology beyond the conventional T1 and T2 MRI. For instance, diffusion-weighted magnetic resonance imaging (DW-MRI) can identify tissues and display their architecture on a microscopic level according to the differences in Brownian motion. [6,7,9] Meanwhile, diffusion tensor imaging (DTI) supplies us with data concerning the directionality of tissue movements and can provide insight into relationships of abnormal borders compared to healthy white matter. Fractional anisotropy (FA) is a scalar value used in diffusion imaging that tells us about the state of white matter myelination, fiber density, and axon diameter. In addition, mean diffusivity (MD) and the apparent diffusion coefficient (ADC) can be used in conjunction with FA to evaluate the integrity of neuronal tissue and assess fiber loss.[7] Magnetic resonance spectroscopy (MRS) can be used to non-invasively provide insight into the body’s biochemical processes. [10]
Another type of imaging technique is the nuclear one which includes positron emission tomography (PET) and single-photon emission computed tomography (SPECT). These provide an image of the brain’s functionality, with SPECT tailored explicitly to detect metabolic processes. PET technology exploits short-lived positron emitters like 11C and 18F, which emit gamma rays that are picked up by the PET scanner to create an image. Although PET imaging is perfect for evaluating brain function, it cannot identify structural abnormalities in the CNS. Moreover, the high baseline metabolic rate of the brain limits the sensitivity of the radioactive glucose used in PET scans for cancer surveillance. Since PET imaging uses X-rays, there is a concern regarding exposure to this ionizing radiation.[7]
Neurosurgery and its goals
Neurosurgical intervention has been a vital tool in the management of CNS tumors. It currently serves three purposes:
- Acquiring a tissue for diagnosis and developing a treatment plan catered to the patient’s condition.
- Partial or complete surgical resection of the tumor and
- Localized drug delivery.[11]
Recent developments in neuroimaging have enabled the incorporation of these imaging techniques intraoperatively, which permitted the surgical resection of lesions that are in delicate locations. For instance, imaging modalities like magnetic resonance imaging (MRI)-based diffusion-tensor imaging (DTI), functional MRI (fMRI), and MRI-guided electrical targeting and electromyography (transcranial magnetic stimulation) improved preoperative planning by precisely outlining the lesion from major tracts and delicate cortical areas. Furthermore, intraoperative monitoring using direct cortical stimulation can help eliminate the drawbacks of static preoperative imaging, which does not consider the changes in the brain position intraoperatively.
Chemotherapy
Some of the most commonly used drugs in chemotherapy are alkylating agents, which alter the DNA, causing DNA strand breakage and triggering apoptosis.
The most popular drug in this class is Temozolomide (TMZ); it constitutes a major part of the treatment regimen post-surgery for patients with IDH-mutant gliomas WHO grade II or III; it synergy with radiation to maximize glioma cell death. The mode of action of TMZ is DNA methylation, which is lipophilic and small, allowing it to cross the BBB. Despite TMZ being a safe drug, it causes dose-limiting myelosuppression.
Other popular drugs within the class of alkylating agents are chloroethyl nitrosoureas which include lomustine, carmustine, and fotemustine. These are highly lipophilic agents which can cross the BBB. Over the years, the use of carmustine in clinical practice has diminished and almost disappeared because of the lung toxicity and prolonged bone marrow suppression it induces. Lomustine is orally administered but causes thrombocytopenia and dose-limiting pulmonary toxicity, while fotemustine causes low pulmonary toxicity but prolonged thrombocytopenia. In general, chloroethyl nitrosoureas induces more leukopenia and thrombocytopenia than TMZ, so patients taking these drugs must be closely monitored.
Sometimes more than one drug can be used together in a polychemotherapy protocol. This is done to avoid cell resistance and amplify the agents’ cytotoxic effects.[3]
Radiotherapy
Radiation is the standard treatment for brain metastases because the BBB allows very few agents to cross it.[12] Multiple brain metastases are typically treated by radiotherapy, which has two approaches, either as whole-brain radiation therapy (WBRT), where it is administered in multiple sessions over 1-3 weeks, or as stereotactic radiosurgery (SRS), where larger doses of radiation are targeted at smaller volumes. Despite clear guidelines on when to use SRS, studies promote its usage in patients with four or fewer lesions. Surgical resection or SRS could be used in patients with a single brain metastasis; the decision depends on many factors, like the lesion size, symptoms, comorbidities, age, and the histopathology report.[11]
Imaging combined with radiotherapy allowed us to modulate the therapeutic window by precisely delivering the radiation. Examples include external beam fractionated stereotactic radiotherapy, radiosurgery, image-guided radiotherapy, intensity-modulated radiotherapy (IMRT), particle therapy, three-dimensional brachytherapy, and intraoperative radiotherapy.[13]
Laser-induced thermal therapy (LITT)
Laser-induced thermal therapy (LITT) is a neurosurgical technique that was developed several decades ago but has recently witnessed a rise in popularity in terms of utilization.[11] The concept of this technique is rather simple; light energy emitted by laser optical fibers is targeted at a tissue, and tumor cells chromophores from this tissue absorb the emitted photons, which causes them to get excited and release thermal energy.[14] This thermal energy diffuses throughout the tissue (thermal diffusion) and causes damage through 3 different mechanisms depending on the amount of thermal energy in the corresponding section. The three mechanisms are laser-induced coagulation (LIC: > 60 °C), dynamic thermal reaction (RDR: 48–60 °C), and laser-induced hyperthermia (LIHT: 42–47 °C).[15] Laser-induced coagulation causes protein denaturation, cellular necrosis, and tissue coagulation, hence inducing irreversible damage, while sections at (42–60 °C) suffer reversible damage. [14,15]
In terms of tumors, this region becomes highly susceptible to relapses making chemotherapy an essential adjuvant. Laser-induced thermal therapy has many advantages besides being minimally invasive and non-ionizing. For instance, it grants access to typically inaccessible places by conventional surgery, reduces the undesired damage to the surrounding tissue (especially if used with MRI), and is associated with a functional improvement postoperatively compared to the conventional surgery (the patient experiences less pain, bleeding, and obstruction), quicker recovery time since it is less physically and emotionally taxing and cheaper for the health care providers. On the other hand, it does have some drawbacks, like its limited usage for large and irregular tumors close to large blood vessels and the inability to accurately control the temperature and dose of the photothermal energy [15].
Aptamers
Aptamers are artificial single-strand oligonucleotides that can bind with high selectivity and affinity and modify the activity of proteins based on their tertiary structural interactions. In therapeutic applications, aptamers adopt one of three roles: antagonists, agonists, and carriers of therapeutic moieties. Antagonists block interactions of certain disease-causing molecules while agonists work by triggering the function of their target molecules, and carriers of therapeutic moieties deliver medicinal agents to their target cells or tissues. The interactions between aptamers and their target molecules resemble antigen-antibody complexes; therefore, aptamers were called “chemical antibodies.” However, compared to traditional antibodies, aptamers display low immunogenicity, high stability, easier tissue penetration, and accessibility to chemical modifications.[16]
In 1990, the systematic evolution of ligands by exponential enrichment (SELEX) method was developed, which is an iterative process where a wide diversity of aptamers is tested to identify the aptamers with the desired characteristics.[17]
Many aptamers were especially useful in managing glioblastoma (GBM). Through differential-cell SELEX, Affinito et al. Identified the aptamer A40s, which can incorporate itself in GBM stem cells and precisely deliver miR-34c and anti-miR10b to the stem cell population. These researchers also showed that A40s were able to cross the blood-brain barrier (BBB) and reach the tumor site to bind the EphA2 receptor causing the cessation of tumor growth and reducing its relapse.
Another group of researchers discovered an aptamer, GL43.T, that can target EphB2/3 receptors and inhibit cell vitality, reducing chemotactic serum- and ligand-stimulated cell migration in vitro with no reports of this aptamer working in animal models.
Another study by Wang et al. Reported the performance of a novel aptamer called CL-4RNV616, which can inhibit the proliferation of U87MG GBM cells by targeting EGFR.[16]
In addition to the role of aptamers in therapeutics, they can also be used in the imaging and diagnosing brain tumors. An aptamer labeled with Alexa-488, and TD05-488, can be utilized to identify tumor margins and neoplastic tissue for the sake of diagnosis. Furthermore, radiolabeled aptamers are used with positron emission tomography and magnetic resonance imaging in animal models. For instance, a study by Gu et al. Highlighted using GBI-10, attached to gadolinium-loaded liposomes, to ameliorate magnetic resonance imaging diagnosis.[16]
Recently approved medications
In 2020, the FDA approved five medications that can prove to be helpful in the field of neuro-oncology. To begin with, NF1 is a rare syndrome that causes individuals to develop peripheral and plexiform neurofibromas, the latter being large tumors that grow from peripheral nerves causing significant morbidity based on their location. Plexiform neurofibromas are typically benign tumors but can transform into malignant peripheral nerve sheath tumors (MPNSTs). On April 10, 2020, the FDA approved the use of selumetinib in pediatric patients (under two years old) with NF1-associated symptomatic, inoperable plexiform neurofibromas. This medication is given orally and inhibits mitogen-activated protein kinase (MAPK) kinase (MEK), which is highly influenced by the loss of the tumor suppressor Neurofibromin 1.
On April 17th, 2020, the FDA authorized the use of Tucatinib in conjunction with trastuzumab and capecitabine for the treatment of advanced unresectable or metastatic human epidermal growth factor receptor 2 (HER2)-positive breast cancer in adult patients, including those with brain metastases.
On May 8th, 2020, Selpercatinib was accepted to be used in the treatment regimen of adult patients with metastatic RET fusion-positive NSCLC. This particular rare type of NSCLC was found to be highly associated with brain metastases.
On September 4th, 2020, Ppralsetinib was approved for use in patients with metastatic RET fusion-positive NSCLC.
On May 22nd, 2020, the kinase inhibitor brigatinib was authorized to treat adult patients with anaplastic lymphoma kinase (ALK)-positive metastatic NSCLC. [18]
Intra-arterial administration of drugs
Over the past few decades, the intra-arterial administration of drugs has gained popularity and showed better overall median survival than its counterparts, especially in treating recurrent or progressive malignant glioblastomas, retinoblastomas, and primary CNS lymphomas.
In this mode of administration, therapeutic agents are directly injected into the carotid or vertebral artery, which causes the concentration of these agents to spike rapidly inside the tumor but, unfortunately, causes severe neural and vascular toxicity. For example, patients with primary glioblastomas or anaplastic astrocytomas being treated with intra-arterial cisplatin typically suffer high-frequency hearing loss. Furthermore, patients who had been recently diagnosed with or even recurrent malignant gliomas experienced decreased visual acuity and irreversible encephalopathy when they were treated with intra-arterial carmustine 1,3-Bis(2-chloroethyl)-1-nitrosourea (BCNU). Additionally, patients with primary CNS lymphomas who were administered intra-arterial nitrosourea 1-(4-amino-2-methyl-5-pyrimidinyl)methyl-3-(2-chloroethyl)-3-nitrosourea (ACNU) in combination with radiotherapy developed myelosuppression. However, the most commonly documented complications were nausea, vomiting, thrombocytopenia, seizures, ocular pain, headache, intratumoral hemorrhage, transient cerebral ischemia, granulocytopenia, nephrotoxicity, and vasospasm.
Specific techniques were developed to enhance the therapeutic effects of intra-arterial drug administration. The five most important ones will be discussed:
The use of chemical agents (like as mannitol, bradykinin, or alkylglycerols) or penetration drug carriers (like liposomes, micelles, and cell-penetrating peptides) to disrupt the blood-brain barrier, which helps increase the entry of drugs into tumor and brain tissues.
Infusing the therapeutic agent intra-arterially during transient cerebral hypoperfusion (IA-TCH) or flow arrest (IA-FA) helps raise the local plasma drug concentration and exposure time by minimizing dilution, absorption, and contact with blood components.
Employing specially fabricated microcatheters to permit super-selective intra-arterial cerebral infusion (SIACI) directly into the arteries supplying the tumor decreases neurotoxicity intensity while providing well-targeted delivery.
The utilization of imaging techniques like MRI, CT, X-ray, SPECT, and PET in conjunction with intra-arterially administered drugs allows the drug delivery and accumulation in the tumor to be monitored.
Adding other tools like gene-edited viruses or cells to intra-arterial administration helps focus the therapy on tumor cells.[19]
Corticosteroids
Corticosteroids are a class of anti-inflammatory drugs that are commonly included in the treatment regimens for cancer. The reason for that can be attributed to three main properties that they possess. These hormones alter the permeability of the blood-brain barrier, which is often impaired due to the cytokines (like vascular endothelial growth factor (VEGF)) secreted by brain tumors. They modulate the permeability through several genes and molecules, including claudins, occludin, zona occludens (ZO)-1, and vascular endothelial (VE)-cadherin, all of which affect the permeability of the endothelium. This results in the decrease of the blood-brain barrier permeability and the extravasation of fluid, reducing edema.
Moreover, corticosteroids can cause the cessation of the cell cycle and, therefore, cell death primarily through apoptosis in a p38 mitogen-activated protein kinase (MAPK)-dependent manner in B and T cells, which makes them effective in the treatment of primary CNS lymphomas (PCNSLs) or secondary lymphomatous neoplasms at the beginning of the treatment process. The effect of corticosteroids against lymphomas is transient, meaning the tumor must be treated by chemotherapeutic agents or irradiation to avoid relapsing.
There is no clinical evidence regarding gliomas that steroids are effective in their treatment. In fact, some preclinical findings show that glucocorticoids make tumor cells resistant to chemotherapy.
Finally, some of the most common side effects in all chemotherapeutic agents are nausea and vomiting, so certain corticosteroids like methylprednisolone and dexamethasone have been used to treat Chemotherapy-induced nausea and vomiting (CINV). Dexamethasone can also be combined with a 5-HT3 receptor antagonist and aprepitant as prophylaxis in patients treated with highly ematogenic agents. On the other hand, corticosteroids have side effects that depend on the type of agent used and its dose. Some of these complications include arterial hypertension (due to hypervolemia), osteoporosis, steroid-induced diabetes, steroid-induced myopathy, psychiatric effects (like insomnia, depression, hypomanic and manic episodes), cataracts, avascular bone necrosis, immunosuppression and therefore opportunistic infections.[20]
Prognosis
Studies have shown that a few factors affect the prognosis of the patient by varying degrees. To begin with, age, tumor grade, preoperative KPS (Karnofsky Performance Scale, a tool that measures the patient’s functional status), surgical method, postoperative radiotherapy and chemotherapy, and postoperative TMZ treatment were all significant prognostic factors.[21,22] Out of these determinants, chemotherapy, the use of TMZ, and age were the most influential, with radiotherapy, KPS, and surgical method having less influence but also being crucial. Conversely, tumor grade, gender, and tumor size were shown to have little effect.[22] References...