Article topic: Glioblastoma Multiforme (GBM)
Author: Randa Shahwan
Editor: Hamza Aljarrah, Rahmeh Adel
Reviewer: Dr Adam M. Abdallah
Keywords: glioblastoma, GBM, astrocytoma, glioma, brain tumor.
Abbreviations: GMB: Glioblastoma Multiforme, MRI: Magnetic resonance imaging, FLAIR: fluid-attenuated inversion recovery, PET: Positron emission tomography.
Abstract
Glioblastoma multiforme (GBM) is a very aggressive and incurable malignant primary brain tumor, with high morbidity and mortality rates, which make it known for its poor prognosis. It is a grade IV astrocytoma. GBM is relatively rare, however, it’s the most common primary malignant brain tumor in adults. Signs and symptoms of GBM vary greatly, but common symptoms are headache, seizures, neurological deficits, and cognitive impairment. It’s often diagnosed by different modalities of neuroimaging, such as MRI and CT scan, along with biopsy. Treatment of GBM is typically multidisciplinary, including surgical resection, radiotherapy, and chemotherapy.
Introduction
The nervous system is made of two main types of cells: neurons and glial cells. Neurons are electrically excitable cells that generate and transmit action potentials to different parts of the body. Glial cells support neurons structurally, they provide neurons with nutrients and protection, they form myelin, and they maintain homeostasis.
While neurons are the basic functional unit of the nervous system, glial cells are more numerous than neurons. There are 6 major types of glial cells (Figure 1) in both the central nervous system (CNS) and the peripheral nervous system (PNS), which are: astrocytes, ependymal cells, oligodendrocytes, microglia, satellite cells, and Schwann cells. Each type of glial cell has its specific function. In this article, we’ll focus on astrocytes.
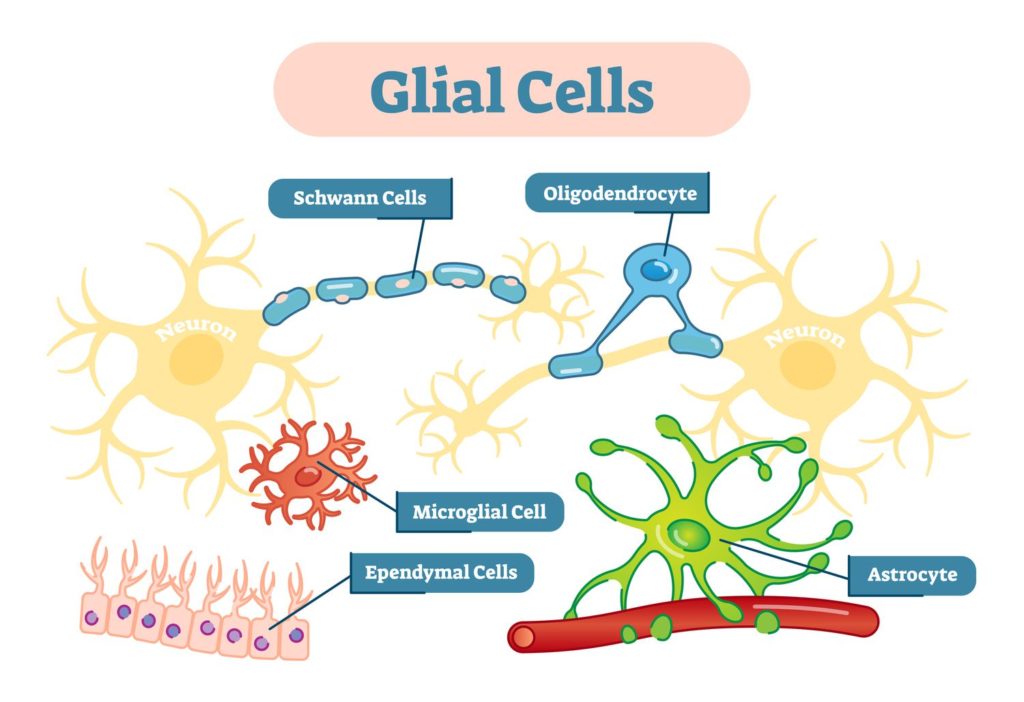
Figure 1: There are 6 major types of glial cells in both the central nervous system (CNS) and the peripheral nervous system (PNS). 15
Astrocytes are considered to be the most numerous glial cells in the CNS. Astrocytes’ roles include structural and metabolic support, maintenance of the blood-brain barrier (BBB), regulation of cerebral blood flow, and inflammatory reactivity after injury.1
When a tumor originates from any type of glial cells, it’s considered a “glioma”. Based on the specific cell type from which the tumor originates, there are three major types of gliomas: astrocytoma, ependymoma, and oligodendroglioma. Considering their growth pattern and infiltrating capacity, gliomas are classified according to the World Health Organization (WHO) by Grades (I-IV). The grade a tumor has is correlated with its aggressiveness.2
Definition
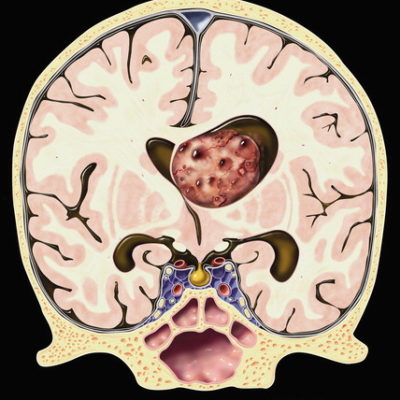
Glioblastoma multiforme is an astrocytoma and a grade 4 glioma based on the (WHO) grading system. Although it is the most common primary malignant brain tumor in adults, it’s a rare malignant tumor overall. It is also the most aggressive primary malignant brain tumor.3 Although it’s highly aggressive, generally, it rarely metastasizes. It also rarely occurs in children. It’s more common in males than females.4 Generally, it has a very poor prognosis and is rarely cured.3 It most commonly forms in the frontal lobe, followed by the temporal, parietal, and occipital lobes.3
Risk factors
The exact cause of GBM is unknown. However, there are some risk factors related to GBM. Risk factors found to be associated with GMB include some genetic disorders, such as Li-Fraumeni syndrome, neurofibromatosis, and Lynch syndrome; previous radiation therapy;5 and impaired immunity.6
It is suggested that in some cases, there’s a genetic component, which in turn, may run in families.4 Most commonly, GBMs arise de novo, but in rare cases, a lower-grade brain tumor can transform into a GBM.7
Clinical presentation
Symptoms of GBMs are nonspecific. The clinical presentation of GBM varies greatly depending on the location of the tumor, its size, and the anatomic brain structures infiltrated by the tumor.3
GBMs often cause symptoms due to mass effect, or increased intracranial pressure, these symptoms include headache, nausea and vomiting, change in personality, slowly progressive focal neurologic deficits, double or blurred vision, depression, and loss of appetite.3
Diagnosis
The diagnosis of GBM relies on a full medical history and physical examination -focusing on neurological examination to check if there’s any neurological impairment- imaging modalities including MRI, CT,3 and PET scan9, and biopsy.
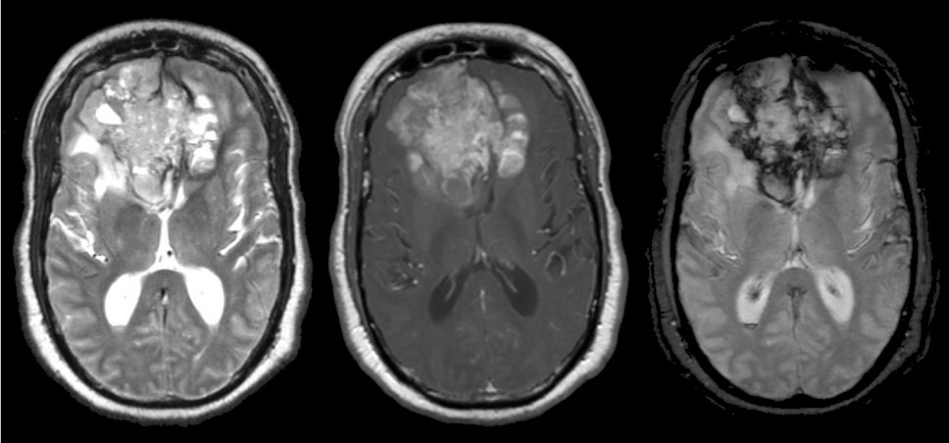
Magnetic resonance imaging
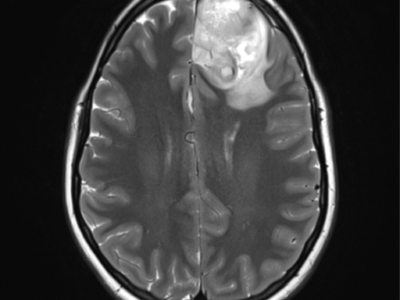
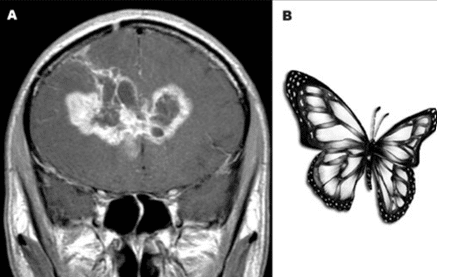
MRI with and without contrast is considered the gold standard in the diagnosis of GBM. On MRI (Figure 2), GBM appears as a poorly circumscribed mass with contrast enhancement at its margins. The center of the mass appears hypointense on T1-weighted scans. It’s surrounded by edema which looks hyperintense on T2-weighted and FLAIR scans.8
Biopsy
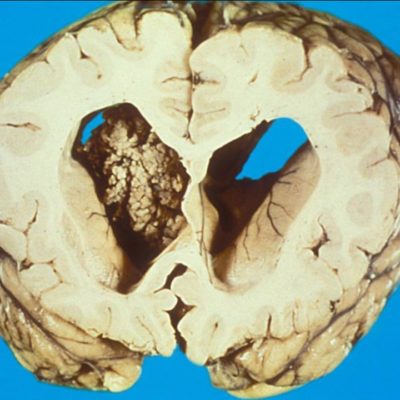
A biopsy mostly confirms the diagnosis. GBM is characterized histologically by the hypercellularity of bizarre giant cells with marked atypia, numerous mitoses, microvascular proliferation, and necrosis.10
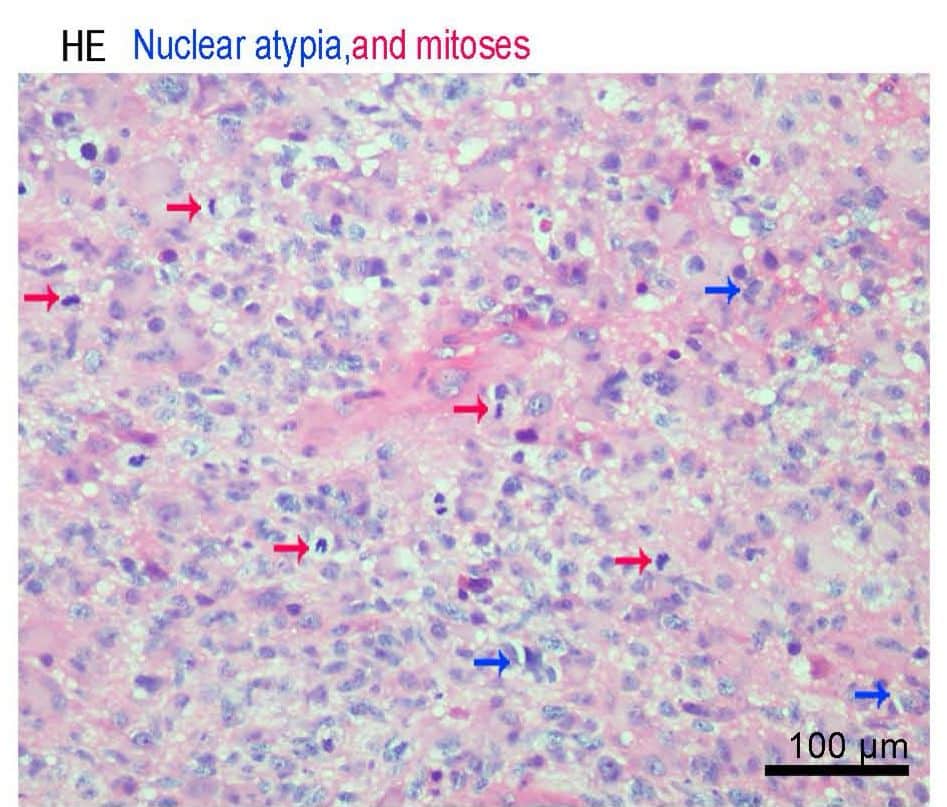
Figure 3: Nuclear Atypia and Mitosis 17
a. Open surgical biopsy
b. Stereotactic biopsy
It is considered a minimally invasive, safe and accurate procedure. It is preferred if the lesion is deep, or critically hard to reach. It is also done when other procedures are considered risky for the patient.16
Biopsy techniques:
1. Core Needle Biopsy
2. Fine Needle Aspiration (FNA)
Prognosis
GBM has a 7.2% 5-year survival rate.11 Without therapy, GBMs have a median survival of 3 months. If managed with optimal management, they have a median survival of approximately 15 months.4 Primary GBMs generally have a worse prognosis than secondary GBMs.3 Overall, GBM has a very poor prognosis, whether treated or not.
Treatment
If glioblastoma is recently diagnosed, it must be managed with a multidisciplinary approach. There are several treatment options currently, which include surgical resection, chemotherapy, and radiation.3 Standard therapy involves maximal safe surgical resection, then radiotherapy concomitant with temozolomide (an oral chemotherapy medication), followed by adjuvant chemotherapy with temozolomide.3 This combination of radiotherapy and chemotherapy is known as the Stupp regimen. However, in older patients, shorter courses of radiotherapy, or only chemotherapy with temozolomide may be considered. After the radiation therapy, chemotherapy with temozolomide is done.3
A radical resection of glioblastoma is not curative, because it is a highly invasive tumor, so most likely, infiltrating tumor cells will remain within the surrounding brain tissue.12
New treatment modalities such as tumor-treating fields and immunotherapy options are currently under trial.3 Immunotherapy options against GBM include dendritic cell vaccines, peptide vaccines, checkpoint inhibitor therapy, chimeric antigen receptor T cells, and oncolytic virotherapy.13
Unfortunately, despite all treatment options available, until now, GBM remains incurable.12
References...