Title : Arachnoid Cyst
Author : Rania Sanouri
Key Words : Brain Cyst , Pediatrics , Seizure , VP Shunt
Scientific Editor : Dr Omar Jbarah
Introduction:
Arachnoid cysts are benign fluid collections found within the arachnoid mater layer of the meninges (1). The first reported cases of IACs were in 1831, in which the majority (75%) of the cysts were found in children (2). Population studies revealed that arachnoid cysts makeup about 1% of intracranial space-occupying lesions and are found in 1.7% of the adult population and 2.6% in the pediatric population (1). Most arachnoid cysts are incidentally discovered with only a minority of the cysts can become symptomatic and require a neurosurgical intervention (2). The etiology behind their presence is poorly understood and usually seen when workup for headache, increased head circumference, or developmental delay is ordered. Most arachnoid cysts can be found in congenital anomalies. Arachnoid cysts could arise secondary to traumas, hemorrhages, or meningitis. It is infrequent for these cysts to cause cranial neuropathies, seizures, or psychiatric disorders (3)(4)(5).
Epidemiology:
Intracranial arachnoid cysts (IACs) have a male to female predominance of 2:1 to 5:1(2). They can occur in association with many genetic syndromes including AD polycystic kidney disease, Neurofibromatosis, mucopolysaccharidosis, schizencephaly, Down’s syndrome, acrocallosal, and Aicardia syndromes (2). Acquired arachnoid cysts represent a smaller number of cases compared to congenital cysts (6). Symptomatic IACs incidence as reported in a large pediatric series is around 6.8% (2).
IACs arise throughout the neuroaxis and half of the cysts are in the middle temporal fossa (MTF). Other locations are the posterior fossa, the suprasellar region, and rarely intraventricularly (2). Middle fossa or Sylvian fissure cysts are the most common in all age groups (7).
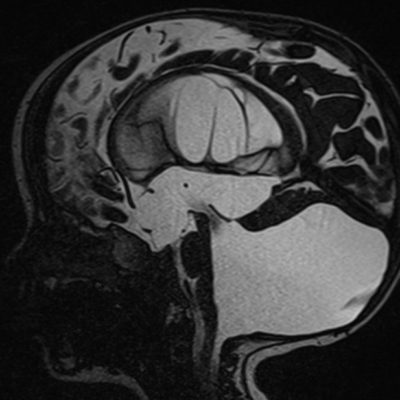
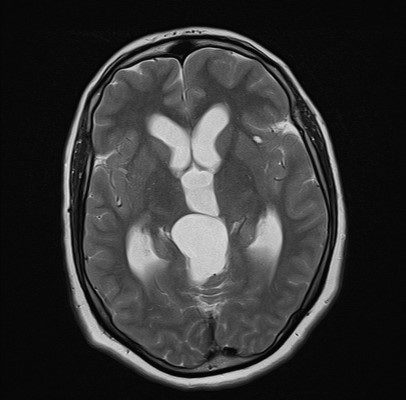
Amongst adults, middle fossa and retro cerebellar cysts show almost similar prevalence (34% and 33%, respectively) (7). Middle fossa cysts in adults were associated with a lower rate of symptoms, whereas cysts located in the cerebellopontine angle, quadrigeminal cistern( Figure 1), sella, and ambient cisterns, were found to be symptomatic. (7).

Figure 1: Quadrigeminal cistern, Case courtesy of Dr Farhad Farzam (63)
Pathogenesis:
Since most cases are seen in the pediatric population, a congenital or genetic cause is suspected (8). Various hereditary syndromes, such as Marfan’s syndrome, neurofibromatosis, glutamic aciduria type I, autosomal dominant polycystic kidney disease, and tuberous sclerosis, were proved to have a higher incidence of arachnoid cysts than the general population (9). A recent genetic association was defined in a consanguineous family with a high prevalence of arachnoid cysts. A link was found between familial arachnoid cysts to chromosome 6q22.31 -23.2.
Earlier, IACs were thought to result from gestational ischemic, traumatic, or infectious insults; these theories are no longer supported (2). A more recent viewpoint for middle fossa IACs is that it was a failure of the frontal and temporal embryonic meningeal merging, resulting in a duplication within the Sylvian fissure (2). Some suggestions about IACs origination include that It is being achieved by splitting of the arachnoid membrane layers during fetal development, forming a “potential space” for cerebrospinal fluid (CSF) (2).
Embryology:
The origin of arachnoid cysts is still unclear and has many controversies. Their formation is believed to be the result of abnormal embryologic development of the subarachnoid space. (11)
At early normal embryonic development, one layer of connective tissue called the meninx Primitiva, or “the perimedullary mesh” will be the precursor to the pia and arachnoid maters and line the surface of the dura and surround the neural tube. At 15 weeks of gestation, the rhombic roof will rupture, and CSF leaves this mesh, pia and arachnoid matters will incompletely separate, resulting in the cobweb-like appearance of the arachnoid. (11)
The primary hypothesis for arachnoid cysts formation implies that separation of the superficial arachnoid and pia matter is aberrant and that enclosed chambers form and develop into a cystic mass. Instead, the meninx Primitiva formation is aberrant, which may also lead to cyst formation. This hypothesis was found to be accepted, as arachnoid cysts were near an arachnoid cistern in most of the cases. (11)
This embryologic model explains the presence of cysts but lacks an explanation of how and why these cysts expand. Numerous explanations were proposed about why cysts may generate intra-cystic pressure and produce parenchymal compression, though, most were unsatisfactory and no direct evidence of fluid production within the cyst has been found. (10)
According to the size, most cysts remain stable in size and rarely disappear, moreover, a slit valve may exist between the subarachnoid space and the cyst, allowing only entry of CSF. Slit valves are directly being observed with endoscopy and until this time, are the most direct explanation for why the cysts expand. (12) (13) (14)
CT cisternography or cine-phase contrast-enhanced MRI sequences usually show filling or emptying of the arachnoid cyst. On normal CSF pulsation or Valsalva maneuver, in addition to the geometry of the split membrane, the pressure gradient which drives fluid into the cyst can be assessed. (12)(13)(14)
Clinical presentation:
Diagnosis of IAC was found more common in childhood, as the prevalence is most at 1 and 5 years (3.8% and 4.6% respectively). (3) This correlates well with previous studies that found children younger than 2 years had higher prevalence. (15)
The clinical presentation for non-incidental paediatric arachnoid cysts is very similar to other childhood mass lesions. The most common indications for work-up and thus treatment of arachnoid cyst involved progressive macrocephaly, intracranial hypertension, headache, hydrocephalus and developmental delay. (3) (15)
The most common complaint found in adult patients was headache, However, other complaints in symptomatic arachnoid cysts involved vertigo, ataxia, hearing loss and seizures. (also common) 2
It was found that arachnoid cysts predispose patients to intracranial hemorrhage for example after mild head trauma. (16)
One theory regarded by computational mechanistic study, supports the fact that hemorrhage is attributed to the elevated sheer forces trauma that is delivered to the outer edge of the cyst relative to the normal brain. (17)
It also states that larger cysts are more likely to cause hemorrhage. According to Cress and his colleagues, 6% of arachnoid cysts were associated with hemorrhage over a 5-year period. Factors that highly increase the risk for hemorrhage include large maximal diameter of the cyst in addition to history of head trauma. (18)
Wester and Helland stated that 4.6% of arachnoid cysts over 17 years and noted that hemorrhages occurred more in temporal cysts. (19) In this study group, only 6% with temporal cysts were found to have cyst related hemorrhage. Both studies assume that cyst-related hemorrhages are rare events.
The overwhelming majority of IACs do not change in size from the time of the initial diagnosis. These lesions only rarely expand in the pediatric population; when they do occur, they are primarily found in children under 4 years of age. (2)
The following table describes the wide range of presenting symptoms:(Table 1)
Table 1 (2)
Range of presenting symptoms
| Symptoms | Mechanism/Notes | |
|---|---|---|
| Symptoms attributable to mass effect either from the IAC, from cyst rupture, or from cyst hemorrhage | Symptoms of increased ICP (HA, emesis, AMS, vision changes, etc.) | Can be obstructive HCP from compression at a variety of places or nonobstructive HCP presumed to be due to abnormal SCF flow dynamics |
| CPA syndrome (tinnitus, hearing loss, facial palsy, nystagmus, and/or vertigo) | CPA IAC | |
| Parinaud’s syndrome | Tectal compression by quadrigeminal plate IAC | |
| Glacial change (bony remodeling) with or without proptosis | Typically, from MTF IACs resulting in cranial asymmetry ranging from focal skull prominence to disfiguring deformity with distortion of the nearby orbit with proptosis | |
| Endocrinopathy | Compression of HPA structures by suprasellar IAC | |
| Seizures | Epileptogenicity localized to affected cortex | |
| Nonspecific symptoms with unclear relationship to IAC | Headache | Headaches unable to be attributed to increased ICP |
| Nonlocalized seizures | Seizure foci unable to be precisely localized to affected cortex | |
| Developmental delay or cognitive deficits | Controversial |
IAC=Intracranial arachnoid cysts; ICP=intracranial pressure; CPA=cerebellopontine angle
Supratentorial Arachnoid Cysts
The most common type of intracranial cysts is Sylvian fissure arachnoid cysts or “middle or temporal fossa cysts”. Almost half of the cysts that occur in pediatrics are found in the middle fossa, and predominantly in the male gender on the left side (66%) (3). In adults, IACs are less prevalent (34%) and their distribution on both sides is the same as in children (7) No statistical significance outside the temporal fossa was described in gender differences. (20)
The most common presentation is a unilateral headache in the supraorbital or temporal region that may be increased with physical exertion. The second most common symptoms are Focal, complex-partial, or generalized seizures. These seizures occur in up to one-third of the patients. It is rare to find symptoms of Increased intracranial pressure (ICP) like nausea, vomiting, or papilledema. (21)
Galassi classification (1982) is an extensively used classification for middle temporal fossa (MTF) IACs, which account for half of all IACs. It classifies MTF cysts into 3 significant types based on their sizes and the mass effect they exert. (2) Type 1 cysts are small lens-shaped collections at the anterior pole of the middle fossa, they cause little mass effect and are not associated with calvarial deformities. They freely communicate with CSF on MR-cine or CT-ventriculography. Type I cysts are the most common type, as 68% of the cysts are in this category and rarely require surgery. (2)(3)
Type II cysts are intermediate in size with a rectangular morphology extending to Sylvian fissure they exert a local mass effect on the temporal lobe and cause midline shift, thus requiring surgery. (2)
Type III cysts are large, rounded cysts that involve the entire length of the Sylvian fissure. They usually cause midline shift due to marked mass effect with thinning and expansion of the middle fossa cranial bones (thinning of temporal squama or displacement of the wings of the sphenoid bone) or spreading of cranial sutures in young children. Recent studies found 17% of the arachnoid cysts to be found in this category. They occupy the middle fossa and extend into the anterior fossa compressing the frontal lobe. These cysts do not communicate with the subarachnoid space and usually require surgery. (2)(3)
Parasellar IACs are subdivided into intrasellar and suprasellar cysts according to their relationship to the diaphragma sella. 50% of supraceller cysts were diagnosed in children less than 5 years and 20% less than 1-year-old. Symptomatic supracellar cysts cause hydrocephalus when they reach the third ventricle and obstruct the aqueduct of Sylvius. Other serious symptoms may include visual abnormalities, gait ataxia, opisthotonos and a syndrome called “Bobble-head doll” syndrome.10-60% of suprasellar cases are going to have endocrinopathies including precocious puberty and growth hormone deficiency. (22) (23)
However, according to many MRI studies, a finding of a suprasellar cyst was not significantly associated with the presence of symptoms. (3)
Infratentorial Arachnoid Cysts
Arachnoid cysts in the cerebellopontine angle usually cause symptoms very similar to Meniere’s disease, those include tinnitus, vertigo, facial weakness, hearing loss and ataxia. Other symptoms are trigeminal neuralgia and hemifacial spasm. (24)(25)(26)
One entity of IACs presents as a large posterior fossa cyst in the midline, and usually cause an obstructive hydrocephalus. These cysts should be distinguished from other lesions seen in Dandy-Walker malformation, mega cisterna magna, epidermoid cysts, large cystic tumours. (1)
It is unusual for posterior fossa cysts to present with cerebellar signs like nystagmus, ataxia, and cranial nerve dysfunction or even progressive quadriparesis. (27)(28)(29)
Imaging
Congenital arachnoid cysts on CT or MRI appear as well circumscribed, smoothly marginated, nonenhancing lesions that are isodense and intense to CSF. They have relatively thin walls that exert mass effect on the surrounding structures, and borders that are difficult to be seen on conventional Imaging. The mass effect on adjacent structures and associated anomalies can best be visualized when combined with contrast imaging to highlight the surrounding vascular anatomy.
Ultrasound often identifies arachnoid cysts in utero or in the neonatal period, but more advanced imaging is usually needed for further characterization.
A noncontrast CT is used to differentiate an arachnoid cyst from other cystic lesions. Communication between the cyst with the basal cistern and the subarachnoid space can be appreciated by CT ventriculography or (cisternography). (27)(28)(29) CT alone is not enough for demonstrating CSF flow dynamics and communication of the arachnoid cyst to the surrounding subarachnoid space.
CT with intrathecal or intraventricular injection of metrizamide can give anatomic detail of the subarachnoid and intraventricular space.
In noncommunicating cysts, it takes 2 to 6 hours for the contrast(metrizamide) to enter inside the cysts, thus the contrast accumulates around the cyst in the cisterns first, creating a halo demarcating the cyst boundary. The contrast then clears from the subarachnoid space and accumulates within the arachnoid cyst.
In communicating arachnoid cysts, the cyst fills with metrizamide, but It’s clearance from the cyst is delayed with respect to the surrounding subarachnoid space and basal cistern.
MRI has a high resolution to detect even the smallest cysts and those found near bony structures. 3D relationship of arachnoid cysts to the basal cistern can be seen with multi-planar acquisition of MRI.
MRI can distinguish between CSF-like fluid of most arachnoid cysts and the proteinaceous fluid of other cystic lesions.
MRI sequences can differentiate epidermoids from arachnoid cysts by the hyperintense appearance on proton density–weighted and diffusion-weighted sequences. (30)
Cysts caused by parasitic infections also appear hyperintense on proton density–weighted images. (31)(32)
Treatment
Conservative Management
Natural history tends to favor conservative management for arachnoid cysts. With the increasing demand for central nervous system imaging, it is likely that the incidence of arachnoid cyst diagnosis has increased as well. Although headache is the most common presenting symptom, it is far from the only complaint suggestive of a symptomatic cyst. Additionally, confounding treatment decision making is the observation that even though headache is a common medical complaint, arachnoid cyst is an uncommon finding. Correlation of these common symptoms to pathology does not necessarily imply causation. Therefore, a detailed neurological and psychiatric history is important in the diagnosis and evaluation of patients with arachnoid cysts. It is also worth noting that conservative treatment has been applied successfully to symptomatic cysts or enlarging cysts as well. (33)(34)
Although there is considerable controversy about what treatment should constitute the standard of care with regard to arachnoid cysts, survey results indicate that most surgeons prefer observation over direct surgical intervention. A study by Tamburrini and associates assessed the practice patterns of the international pediatric neurosurgery community through a case history survey. (35) Participants were asked to make recommendations in the case of a pediatric male patient found to have an asymptomatic Galassi type II arachnoid cyst. Most (60%) recommended continued clinical follow-up, and 50% recommended a baseline MRI. A minority (17%) recommended surgery to reduce the lifetime risk for hemorrhage, and 26% recommended avoidance of contact sports for the untreated cyst. (3)
Arguments against conservative management tend to rest on the subtlety of symptoms as well as the risk for cyst hemorrhage or expansion. As stated previously, the risk for hemorrhage is relatively minor (approximately 5% of patients), although no prospective trial has assessed the lifetime risk in these patients. The risk for cyst expansion has been similarly shown to be low. In a subgroup analysis of 111 pediatric patients who presented with what were thought to be asymptomatic arachnoid cysts, Al-Holou and colleagues found cyst expansion in 11 patients, decrease in cyst size in 13 patients, and stable cyst size in the remaining. (3)
Of those 11, 4 eventually underwent surgery because 3 developed symptoms related to the cyst and 1 had concomitant intractable temporal lobe epilepsy. The authors also noted that cyst expansion seemed to occur exclusively in the very young patients; no patients who were found to have arachnoid cysts after 4 years of age experienced cyst expansion. (35)
Surgery
Arguments in favor of surgery tend to cite the improvement in the wide variety of symptoms as the rationale for intervention. Other potential consequences of intracranial mass lesions, such as intracranial hypertension, have been investigated as well, but the results have not helped to identify those patients who will benefit from surgery. (35)(36) Di Rocco and colleagues demonstrated that the 2 patients in the study with Galassi type III cysts had universally elevated ICP on invasive monitoring.(35) Half of the patients with Galassi type II cysts had increased ICP, but the implications of this finding were unclear because these patients had cysts that were either asymptomatic or had been diagnosed prenatally. When assessing the effect of elevated intracystic pressure on preoperative symptoms or postoperative results, Helland and Wester found a positive correlation between pressure and the severity of symptoms. (36) However, the mean intracystic pressure was within normal limits for all types of temporal cysts. Patients from all groups experienced a postoperative improvement in symptoms, and this improvement was independent of the improvement in cyst size. These studies show that the mechanism of symptomatology may be more complex than simple pressure differentials.
In terms of surgical approaches, several techniques have been shown to be effective for arachnoid cyst treatment. For pediatric patients, a meta-analysis of reported case series revealed similar quality-of-life outcomes for all three types of intervention. (37)
Cystoperitoneal Shunt
Some advocate placement of a cystoperitoneal shunt as a first-line treatment to achieve early cyst obliteration while avoiding large fluid shifts associated with microsurgery. (37),(38),(39) This practice is controversial, however. Cystoperitoneal shunting may offer the advantage over microsurgical fenestration for improved reduction in postoperative cyst volume (74% versus 58%).(40) Risks
unique to shunt placement include shunt dependency, slit ventricle syndrome, shunt infection, and requirement for shunt revision.
Strategies to avoid these complications include placement of an adjustable pressure valve, attempted removal when cyst obliteration is demonstrated, and avoidance of low-pressure or
valveless shunts. (41-43) In centers where shunting is the preferred first-line treatment for arachnoid cysts, authors report similar efficacy to microsurgery and similar rates of shunt complication to other pathologies treated by shunting. Placement of a ventriculoperitoneal shunt may be necessary to treat concomitant hydrocephalus before resection of the cyst. Finally, for patients presenting with quadrigeminal cistern or pineal region arachnoid cysts, Torkildsen shunt variation95 offers the ability for cyst drainage without the risk for overdrainage. (44)
Endoscopic Fenestration
Neuroendoscopy has emerged as an effective alternative to microsurgical fenestration and excision. The surgical strategy for cyst fenestration remains the same, but endoscopy offers several advantages over microsurgery. For midline and intraventricular cysts, endoscopy offers the ability to visualize the boundaries of the cyst as well as critical adjacent structures but reduces the need for extensive dissection. The overall clinical success rate is 83% to 92%. (45-53)
Suprasellar, quadrigeminal, and posterior fossa cysts have higher rates of success compared with Sylvian fissure and interhemispheric cysts. Meta-analysis of published case series demonstrated no
significant difference in rate of success for endoscopic fenestration of suprasellar, quadrigeminal, and posterior fossa cysts versus open surgery with shunt placement, whereas the rates of success for sylvian fissure and the interhemispheric fissure cysts is significantly less effective. (47)
Some case series report higher rates of recurrence or postoperative complications in patients treated with endoscopy. The most common postoperative complications include subdural hematoma, subdural effusion, and cyst recurrence. However, these numbers are highly variable,and it is not clear what factors are associated with complication development. Several technical modifications can be done to improve outcomes. First, a wide fenestration is generally recommended to avoid cyst reclosure. Ventriculocystocisternostomy may produce more durable results compared with ventriculocystostomy .(55,56)
Spinal Arachnoid Cysts
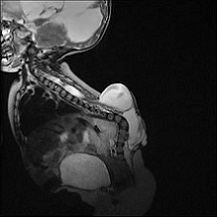
Arachnoid cysts formed within the spine, can either be found intradurally or extradurally. Those cysts usually involve the thoracic spine and cause subtle symptoms mimicking cord compression. (57,58)
Most patients with spinal arachnoid cysts (SACs) complain of intermittent back pain or radicular pain that is exacerbated by Valsalva manoeuvre. According to Wang & his colleagues, cysts that were found ventral to the spinal cord were more to cause myelopathy and weakness, while dorsal cysts caused more numbness. The cause behind IACs is still unknown, but is thought however to be congenital or secondary to infection, trauma or inflammation. MRI is found to be the most sensitive and specific study for diagnosing SACs. SACs can be found incidental just like the case in IACs. (57)(59)
For the purposes of studying fluid dynamics within the cysts, location and differentiation of SACs from other similar lesions, CT myelography, cine-MRI, or diffusion weighted MRI can be done. (58)
Goals of surgical fenestration are similar to IACs. Wide fenestration of the cyst is enough to prevent recurrences. Symptoms of myelopathy and weakness are more likely to resolve after surgery than numbness & pain. Many centers have reported success with an endoscopic approach, either as primary treatment or to look for cyst wall boundaries. (60,61)
Conclusion:
Arachnoid cysts are nonneoplastic lesions that can lead to mass effect and obstructive hydrocephalus. Although most cysts produce no symptoms, some cysts however can cause hydrocephalus, epilepsy, cognitive impairments and even cause psychiatric illness. For symptomatic cases, surgical intervention can be very useful. Microsurgery and endoscopy can aid in decompression of the cysts and CSF circulation restoration, while shunting helps in reducing the size of cysts. Ultimately, Treatment examples are operator dependent and usually give comparable outcomes.(62)
External Links