Article topic: Tourette Syndrome
Author: Amr Atef Hasan
Editor: Said Lahham, Rahmeh Adel
Reviewer: Ethar Hazaimeh
Keywords: Tic, Urge, Dopamine, Obsessive Compulsive Disorder, risk factors
Abbreviations: TS: Tourette Syndrome, CTD: chronic tics disorder.
Abstract
Objective
This article has focused on Tourette Syndrome (TS) and its connection to Obsessive-Compulsive Disorder (OCD), as indicated by a body of genetic epidemiological research that consistently presents compelling evidence of their association. Additionally, the article has provided insights into the risk factors, characteristics, findings from imaging studies, classification, management strategies, and treatment options for this syndrome.
Method
To be considered for inclusion, research papers had to be written in the English language. We conducted searches for relevant articles using databases such as PubMed, Google Scholar, Medscape, and Medline.
Result
Tourette syndrome, a neurodevelopmental disorder, disproportionately affects boys compared to girls and is often associated with attention deficit hyperactivity disorder (ADHD) and obsessive-compulsive disorder (OCD). It can manifest in both children and adults, although it typically begins around the age of 6 years, and adult-onset tics are uncommon. The diagnostic process for TS often includes a clinical Interview, Physical Examination, Diagnostic Criteria, Observation of Tics, Patient and Family History, and Psychological Assessment.
Conclusion
Although Tourette Syndrome is frequently diagnosed in childhood, its precise origins remain a topic of ongoing investigation. Past research has unveiled disparities in the neurobiological makeup of individuals affected by Tourette Syndrome. In recent years, a slew of studies conducted within the last half-decade has brought fresh insights into genetic variations, alterations in neurophysiological processes, and changes in neural networks that contribute to its onset. This has led to a surge in research across various treatment areas related to Tourette Syndrome. Numerous therapeutic approaches have exhibited efficacy in controlled trials, such as interventions involving Dopamine D2 receptor antagonists, Dopamine agonists, Cannabinoids, and Habit reversal therapy.
Overview
Tourette Syndrome is a condition characterized by tics, typically manifesting during childhood and often showing improvement in adulthood1. It represents a chronic neurodevelopmental disorder marked by motor and vocal tics, significantly affecting the quality of life for those it afflicts.2
In 1885, Gilles de la Tourette provided a vivid description of a neuropsychiatric syndrome with distinct clinical features, including multiple motor tics, phonic symptoms, compulsions, and other behavioral phenomena, such as an irresistible urge to echo or imitate. He noted its prevalence in males, early onset, and lifelong duration, speculating that its origins might be linked to inherited neurological vulnerability. Following Tourette’s initial report, over the past two decades, numerous cases and patient series have been documented in the global literature, leaving little doubt that Tourette’s syndrome (TS) is a clinically identifiable disorder with a consistent pattern of expression across different cultures.7
This concept finds support in multiple lines of evidence, including clinical pharmacology, the presence of tics in other disorders characterized by clear basal ganglia pathology, and imaging data indicating developmental abnormalities within the basal ganglia. Additionally, robust backing for the dysfunction of the basal ganglia stems from experimental manipulations of basal ganglia pathways in non-human primates, which produce credible tic-like behaviors.4
Tics observed in Tourette syndrome may be linked to various comorbidities, including anxiety, attention deficit hyperactivity disorder (ADHD), and obsessive–compulsive disorder (OCD). Tourette syndrome and chronic tic disorders (CTD) are among the five categories of tic disorders delineated in the fifth edition of the American Diagnostic and Statistical Manual of Mental Disorders (DSM-5).5
Furthermore, Tourette syndrome appears to cluster significantly within families, implying that family members may share genetic or environmental risk factors contributing to the condition. The morbid risk of Tourette syndrome among relatives ranges from approximately 10% to 15%, while the prevalence of other tics falls within the range of 15% to 20%. These figures notably exceed the rates of Tourette syndrome and tics observed in control groups.6 In this article we will shed light on Tourette Syndrome, with a focus on its definition, etiology, clinical classification, management, and treatment.
Clinical Features
The defining characteristic of Tourette’s syndrome is the persistent presence of at least two motor tics and one vocal tic that have been present since childhood. Tics are described as involuntary, sudden, rapid, recurring, and non-rhythmic movements (referred to as motor tics) or vocalizations, known as vocal or phonic tics. Importantly, this syndrome is now recognized as a global condition, affecting individuals of all races, ethnic backgrounds, and both genders (with a higher prevalence in males, approximately four times more common than in females). Tourette’s syndrome can manifest in both children and adults, although it typically begins around the age of 6 years, and adult-onset tics are uncommon.8
Motor tics generally precede the development of vocal tics, and simple tics often appear before complex tics. Simple motor tics may manifest as actions such as eye blinking, facial grimacing, shoulder shrugging, neck stretching, and abdominal contractions. Among vocal tics, the most frequently observed ones include sniffing, grunting, and throat clearing. Gilles de la Tourette’s original case series featured nine patients who also exhibited complex tics, specifically echolalia (repeating other people’s words) and coprolalia (the involuntary use of profane language). It is worth noting that both coprolalia and copropraxia (involuntary production of offensive gestures as complex tics) are relatively rare, occurring in approximately 10% of patients (with higher rates of 20-30% in specialized clinics that see more severe or complex cases).8
Etiology
Years of investigation have firmly established the influence of genetic factors on Tourette Syndrome (TS). Multiple studies have demonstrated a familial association with TS. Nevertheless, an illuminating twin study revealed that the concordance rate was 53% for monozygotic twins and 8% for dizygotic twins, underscoring that TS is not solely dictated by genetic factors. Consequently, it is understood to be a complex condition arising from a combination of factors, encompassing genetic predispositions and environmental triggers.9
Genetic
Family and twin studies have provided compelling evidence that genetic factors play a significant role in the hereditary transmission of Tourette Syndrome (TS) and related conditions.14 Symptoms of obsessive-compulsive disorder (OCD) are frequently observed alongside Tourette Syndrome (TS), and family studies have indicated a heightened prevalence of OCD among close relatives of individuals with TS. Initial analyses of TS within families suggested an autosomal dominant inheritance pattern with varying expression, encompassing the presence of OCD. However, recent scrutiny has cast doubt on these findings, as observations suggest that the transmission pattern of TS may align more closely with polygenic influences.10
In conditions believed to involve complex genetic inheritance, variations in an individual’s DNA, known as polymorphisms, are thought to heighten the susceptibility to the condition and may contribute to the disease through a cumulative impact. These DNA polymorphisms are commonly referred to as Single Nucleotide Polymorphisms (SNP) and Copy Number Variants (CNV).11
Expanding upon two studies published in 2018, which reported an increased prevalence of rare, gene-disrupting mutations in 5–10% of Tourette Syndrome (TS) patients, notable findings emerged. Wang and colleagues conducted the most extensive examination of rare, de novo gene-disrupting mutations in TS patients to date. Meanwhile, Yu and colleagues conducted the latest TS genome-wide association study (GWAS), involving 4819 cases and 9488 controls. In their study, they identified a strong candidate TS gene (FLT3) and corroborated prior findings indicating that a significant portion of TS heritability can be attributed to the collective influence of common genetic risk variants distributed throughout the genome. These two sets of findings collectively endorse the notion that TS and other tic disorders likely exist on a continuum rather than being distinct diagnostic entities, a viewpoint supported at the genetic level.12
Furthermore, Mufford and colleagues employed the same TS GWAS polygenic risk score (PRS) to investigate genetic data related to subcortical brain volumes in the ENIGMA (Enhancing Neuro Imaging Genetics Through Meta Analysis) consortium. Essentially, this research linked the genetics of TS with the genetics of brain volumes. In a separate study involving a vast dataset derived from shared GWAS data encompassing over a million individuals with various neurological and psychiatric disorders, it was revealed that TS shares common variant genetic risk with conditions such as obsessive-compulsive disorder (OCD), major depression, and, surprisingly, with migraine, particularly migraine with aura.12
In 2021, the TS/OCD working group of the Psychiatric Genomics Consortium conducted a comprehensive genome-wide pathway analysis, involving 1,285 individuals with Tourette Syndrome (TS) and 4,964 ancestry-matched controls. This study revealed significant differences in three gene sets associated with TS: ligand-gated ion channel signaling, lymphocytic (driven by variants in FLT3), and cell adhesion and trans-synaptic signaling sets. These gene sets align with and substantiate previous hypotheses regarding the potential involvement of neuroinflammation, GABA signaling, and cell adhesion mechanisms in the pathophysiology of TS.13
Furthermore, it was recently discovered that a specific genetic variant located on chromosome 13 is linked to Tourette’s Syndrome in a single child. This gene, known as SLITRK1, has been implicated in dendrite growth and the regulation of neural development. However, it’s important to note that only a small percentage of individuals with Tourette’s Syndrome are believed to possess this particular gene mutation.15
Environmental risk factors
The influence of environmental factors on the disease mechanisms and clinical manifestation of Tourette Syndrome (TS) can commence during the prenatal and perinatal phases and persist throughout virtually the entire lifespan of the individual, spanning from childhood and adolescence into adulthood. Exposures to adverse conditions, particularly those occurring early on, such as during pregnancy or due to complications during delivery, may have formative effects on the development of brain networks that are pertinent to the generation of tics and associated symptoms.16
An extensive and authoritative review, of the immunology of brain development in a general context and specifically within the context of Tourette Syndrome (TS), has been authored by one of the leading experts in this field. An intriguing study was prompted by clinical observations that blinking tics, a commonly observed initial symptom of tic disorders, are frequently mistaken for allergic conjunctivitis (AC) by both families and primary care physicians. Researchers in southwest China conducted a study involving 70 children with provisional tic disorder (PTD) and 70 tic-free controls. Their findings indicated that AC was more than four times more prevalent in individuals with PTD (74%) compared to controls (17%). Importantly, this discrepancy could not be solely attributed to symptom confusion, as quantitative assessments of dry eyes and allergic responses to a skin prick test were also about four times more frequent in patients with PTD. These results introduce intriguing hypotheses about immune-related abnormalities that may contribute to the development of tics.15
Additionally, an alternative perspective arises from patient reports indicating that tics sometimes develop after a repeated behavior initiated for a different reason outlasts its initial stimulus and becomes chronic. For example, a child may have initially coughed due to an upper airway infection, but then the cough persisted and transformed into a tic. This suggests that in some cases, tics may emerge when underlying host factors convert externally triggered repetitive behaviors (such as blinking or coughing) into chronic symptoms. Notably, prior research in rodents has demonstrated that a similar two-hit scenario can lead to the development of a distinct movement disorder, dystonia.15
DSM-5 Diagnostic Criteria
The diagnostic criteria for Tourette syndrome, as outlined in DSM-5 (307.23), are as follows:
- Presence of both multiple motor tics and at least one vocal tic at some point during the illness, although they may not necessarily occur simultaneously.
- Tics manifest frequently throughout the day, typically in bouts, occurring almost daily or intermittently for a period exceeding one year. During this duration, there should be no period longer than three consecutive months without any tics.
- The onset of symptoms occurs before reaching 18 years of age.
- The distribution is not attributable to the direct physiological effects of a substance (e.g., stimulants) or a general medical condition (e.g., Huntington’s disease or postviral encephalitis).17
Classification of tics
Tics are conventionally categorized, albeit somewhat subjectively, based on their distribution and complexity. While tics typically manifest in areas such as the face, neck, pharynx, shoulders, and central torso, they have the potential to affect virtually any part of the body (Figure 1).18
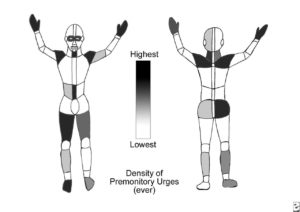
Figure 1: Tourette syndrome and other tic disorders. The graphic shows the relative likelihood of lifetime sensory tics in a given region, as based on self-report of patients with Tourette syndrome. Overt tics are distributed similarly.18
Albin has observed that stimulating the ventral basal ganglia can elicit socially significant complex gestures, such as stereotyped fear or pleasure displays in rodents. Importantly, these socially meaningful gestures predominantly involve the same body regions associated with the ventral basal ganglia.18 However, it’s worth noting that certain tics may not neatly align with traditional classifications.19
Simple motor tics are characterized by brief movements involving a single muscle group, while simple vocal tics involve simple sounds. Some examples of simple motor tics encompass eye blinks, facial grimaces, head and neck jerks, and shoulder shrugs. Additionally, brief sustained postures like blepharospasm, oculogyric movements, and sustained mouth opening can also be categorized as simple motor tics. In the realm of simple vocal tics, you might find actions like coughing, sniffing, throat clearing, grunting, and squeaking. Complex motor tics involve more purposeful or coordinated patterns of movement, whereas complex vocal tics manifest as longer, more organized patterns of speech. Complex motor tics can include actions such as turning while walking, echopraxia (mimicking gestures), and copropraxia (performing obscene gestures). On the other hand, complex vocal tics encompass behaviors like repeating words or phrases, echolalia (repeating another person’s words or phrases), and coprolalia (uttering obscenities).21 It’s worth mentioning that coprolalia, while it can cause significant social distress, is relatively rare, occurring in only approximately 10% of patients.22
General features of tics
The majority of tics are simple, seemingly purposeless movements. However, tics are not entirely random; they often incorporate elements of personal experience, practice, and social norms. For instance, echolalia or echopraxia, which involves mimicking another person’s speech or movements, can occur in up to one-third of patients. It’s interesting to note that witnessing someone with tics, such as excessive blinking, can trigger a similar tic in someone who hasn’t experienced it for years. Likewise, simply hearing or learning about tics can increase their severity. At times, tics appear to imitate newly acquired voluntary behaviors. For example, some individuals have developed a complex tic of “air typing” after learning touch typing, a phenomenon observed in two unrelated individuals by one of the authors. Intriguingly, a man whose personal history was published in 1902 described his tics as frequently involving an urge to engage in precisely what was forbidden, like “clucking” when instructed to remain silent in school. He likened his motor and vocal tics to a “desire for forbidden fruit.19
Complex tics can encompass inappropriate actions, such as touching a hot iron or a stranger’s breasts. As with many movement disorders, tics tend to worsen during times of emotional stress and significantly diminish during sleep.20
Imaging Studies
Structural imaging studies are typically not necessary when evaluating patients with a typical clinical history and examination findings. They are primarily recommended when there are indications of specific illnesses suggested by unusual history or examination results. Currently, functional imaging studies do not have established clinical utility in the assessment of tic disorders. However, there is some potential for future clinical benefits based on data from unpublished reports. For instance, there is emerging evidence that caudate volume in childhood may be inversely associated with the severity of the illness in adulthood.23

Figure 2: Gilles de la Tourette syndrome and voluntary movement: A functional MRI study.35
Management and treatment
Several fundamental principles should guide our approach to Tourette syndrome (TS). Firstly, it’s crucial to acknowledge that current treatments for TS are primarily aimed at alleviating symptoms; there are no known curative or preventive treatments. Secondly, it’s important to recognize that tics, while a prominent feature of TS, may not always be the most distressing aspect of the condition. Thirdly, TS is a chronic disorder, and the focus should typically be on achieving long-term benefits rather than seeking rapid improvement at any cost. Lastly, symptoms can fluctuate, improving or worsening over time, even in the absence of treatment.24
From these principles, several corollaries emerge:
- Treatment may not always be necessary, especially when tics are mild and not significantly impacting the individual’s life.
- Treatment should prioritize addressing the most troublesome symptoms or those causing the greatest impairment.
- It’s important to approach the perceived success or failure of any treatment with caution, as outcomes may sometimes be coincidental.
- Starting with reasonable trials of single treatment agents is generally a more prudent approach than hastily resorting to high doses or using multiple medications.
TS has been categorized as both a neurological and a psychiatric disorder. These labels do not pertain to the cause or treatment of TS but rather reflect the historical involvement of neurologists and psychiatrists in researching and managing the condition.24
Treatment for tics
Effective treatments for tics, supported by evidence from replicated controlled trials (RCTs), include the following:
Dopamine D2 receptor antagonist therapy
In 1959, shortly after its introduction, chlorpromazine was reported to have a remarkable impact on reducing tic severity. Since then, multiple randomized controlled trials, utilizing various neuroleptic medications such as haloperidol, fluphenazine, and pimozide, have substantiated these initial findings.25 On average, treatment with neuroleptics results in a reduction of tic severity by approximately 50-80%. Neuroleptic drugs currently stand as the standard for effectiveness in treating tics. Notably, they can be effective at doses significantly lower than those typically used for treating psychosis, and most adverse effects can be managed through pharmacological adjustments.
Regrettably, some patients find it challenging to tolerate acute adverse effects, which most commonly include sedation, weight gain, depression, lethargy, and akathisia. Additionally, prolonged treatment carries a slight risk of tardive dyskinesia. Consequently, researchers have explored alternative treatment options.26
Dopamine antagonists, such as fluphenazine (Prolixin) at doses ranging from 1 to 15 mg/day and trifluoperazine (Stellazine) at doses ranging from 2 to 15 mg/day, are less commonly used than haloperidol or pimozide but appear to exhibit roughly equivalent efficacy in suppressing tics and share a similar spectrum of side effects. Some clinicians prefer these medications due to their potential to induce less sedation compared to haloperidol.28
Dopamine agonist therapy
Dopamine agonists, which can be categorized into ergoline- and nonergoline DA agonists, have been explored as potential treatments for tics since 1979 when the first positive effects were reported in individuals with Tourette Syndrome (TS).36 Several studies have delved into the impact of dopamine agonists on tics. Despite initial expectations that these agonists might not be effective due to the success of antagonist treatments, some findings have indeed been promising. Both apomorphine and the partial D2 (and 5-HT1A) agonist buspirone have demonstrated efficacy in the treatment of tics. Additionally, ropinirole has been shown to have a positive effect on reducing tic frequency and severity.37
Cannabinoids
High concentrations of cannabinoid receptors have been identified in the basal ganglia and hippocampus, suggesting a potential role of cannabinoids in regulating movement and behavior.34 The cannabis plant is rich in various compounds, with delta-9-tetrahydrocannabinol (THC) being the most abundant and psychoactive among the cannabinoids. Cannabis contains more than 60 cannabinoid compounds, and some, like cannabidiol, may influence the effects of THC.33 Cannabinoids have been utilized for medicinal purposes for centuries. Currently, delta-9-tetrahydrocannabinol (THC) and the cannabis extract nabiximols are approved for the treatment of nausea and anorexia, respectively. In the context of Tourette Syndrome (TS), there have been numerous anecdotal reports suggesting that marijuana may be effective not only in reducing tics but also in addressing associated behavioral issues.27
Botulinum toxin for tics
Local intramuscular injections of botulinum toxin can be considered a therapeutic approach when dealing with a select few highly troublesome tics. Case series have suggested that botulinum toxin injections may not only alleviate tics but also provide relief from associated uncomfortable premonitory sensations and pain. This treatment approach is frequently employed for tics affecting the eye blinking, neck, and shoulder regions. In cases of severe vocal tics, intralaryngeal injections may also be utilized. It’s important to note that the benefits of botulinum toxin therapy are temporary, typically lasting for a duration of 3 to 6 months.29
Habit reversal therapy
Habit Reversal Therapy (HRT) stands as the primary behavioral psychotherapeutic approach for treating Tic Disorders (TD). HRT encompasses three fundamental steps, divided into two stages, and typically spans twelve sessions.3
The three core steps are as follows:
- Awareness Training: This initial step involves guiding individuals, particularly children, to recognize and acknowledge premonitory urges, which are the sensations that precede the occurrence of a tic.
- Competing Response Training: In this phase, specific competing responses are identified for each tic. For instance, for vocal tics, controlled breathing may be selected as a competing response, while for sniffing tics, nose breathing could serve as the competing response.
- Intensive Training: This step involves selecting a family member to assist the child in diligently practicing and reinforcing the competing response, ensuring its effectiveness (Figure 3).
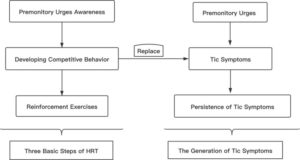
Figure 3: Three basic steps of habit reversal therapy 30
The treatment process is divided into two stages:
Stage One (Sessions 1–8): This stage encompasses four main components: tic awareness training, competing response training, relaxation techniques, and intensive training.
Stage Two: The second stage primarily serves as a review and consolidation phase, ensuring that the learned techniques are effectively applied and maintained.
These sessions are distributed across twelve sessions as follows:
- Session 1: Introduction to the basic process, familiarization with reversal training, and introduction to the methods and procedures.
- Sessions 2–6: Weekly sessions focused on developing distinct competitive behaviors tailored to various tic symptoms.
- Sessions 7 and 8: Biweekly sessions primarily dedicated to reviewing and consolidating the content covered in sessions 2–6.
- Sessions 9–12: Monthly sessions that further reinforce and review the training provided in previous sessions.30
Deep brain stimulation for Tourette Syndrome
Figure 4: Deep Brain Stimulation: A New Option for Tourette Syndrome.32
Deep Brain Stimulation (DBS) has emerged as a viable option for certain individuals with Tourette Syndrome (TS) who experience substantial impairment in their quality of life due to motor and/or vocal tics, even after trying various other treatment approaches.32 (Figure 4)
The current clinical criteria for considering Deep Brain Stimulation (DBS) in a patient with Tourette Syndrome (TS) are primarily rooted in the clinical diagnosis, with an emphasis on high tic severity scores and the persistence of symptoms despite the utilization of at least three different pharmacological drugs. These drugs typically include alpha-adrenergic agonists, dopamine antagonists, and medication from at least one additional class, such as tetrabenazine or clonazepam. While some recommendations may use age as a potential exclusion criterion, particularly limiting DBS for individuals below the age of 18 or 25, it’s important to note that age should not be an absolute or inflexible criterion.32 However, it is advisable to seek guidance and consult with a local ethics committee when contemplating surgery for patients younger than 18 years of age.31
Conclusion
While Tourette Syndrome is a common childhood disorder, its exact cause has not yet been fully understood. Previous studies have revealed differences in the neurobiological structures of individuals with Tourette Syndrome.11 In recent years, research conducted over the past five years has shed new light on genetic variations and changes in neurophysiology and brain networks that contribute to its development.3
Notably, only a limited number of studies have delved into the role of environmental factors in the causation and natural progression of Tourette Syndrome. Nevertheless, the data generated by these studies warrant further investigation in this area.16
Tourette Syndrome disproportionately affects boys compared to girls and is often associated with attention deficit hyperactivity disorder (ADHD) and obsessive-compulsive disorder (OCD). Although the inheritance pattern of Tourette Syndrome frequently appears to be autosomal recessive, pinpointing specific abnormal genes has proven challenging.1 Over the past several decades, treatment options have significantly advanced, offering greater opportunities to enhance the quality of life for individuals with Tourette Syndrome. Promising pharmacological options on the horizon hold the potential to further reduce tics, ideally with fewer adverse effects.3
References